Every year, millions of people undergo treatment that can save their lives, yet the medicine itself carries hidden dangers. When you hear about chemotherapy, your mind goes straight to hair loss or nausea. But there is another side to these powerful drugs. They pose significant risks to the nurses preparing them, the doctors administering them, and even the family members helping care for patients at home. In fact, standard safety practices have evolved significantly over the last few decades, moving from basic advice to rigorous, legally binding frameworks.
The landscape changed dramatically with the release of updated standards in 2024. These rules aren't just paperwork; they represent a major shift in how we protect human life during critical cancer treatments. We are seeing a move away from simply "being careful" toward specific, verifiable protocols that leave no room for error.
The 2024 Antineoplastic Therapy Standards
Antineoplastic Therapy Administration Safety Standards is a comprehensive framework established by major medical organizations to regulate the safe delivery of cancer drugs. The framework was notably updated in 2024 by joint committees including the American Society of Clinical Oncology (ASCO) and the Oncology Nursing Society (ONS). These updates reflect the changing nature of cancer drugs. We used to talk mostly about traditional chemotherapy pills and IVs. Now, immunotherapies and targeted therapies are common, requiring different handling approaches despite similar risks.
The core change everyone needs to know about involves patient verification. For years, checking a patient ID twice was the norm. The 2024 revision mandates a fourth verification step performed right at the patient's bedside. Why does this matter? Because patient identification errors accounted for nearly 18% of adverse events in recent reports. Having two licensed clinicians independently confirm the diagnosis, medication names, doses, and therapeutic goals before the drug enters the body catches mistakes that automated systems might miss.
This isn't just about stopping the wrong drug from reaching the wrong person. It also includes education requirements. Patients must document that they understand the duration of treatment and potential side effects. This ensures informed consent isn't a signature grabbed in five minutes, but a conversation documented clearly.
Essential Protective Gear and Engineering Controls
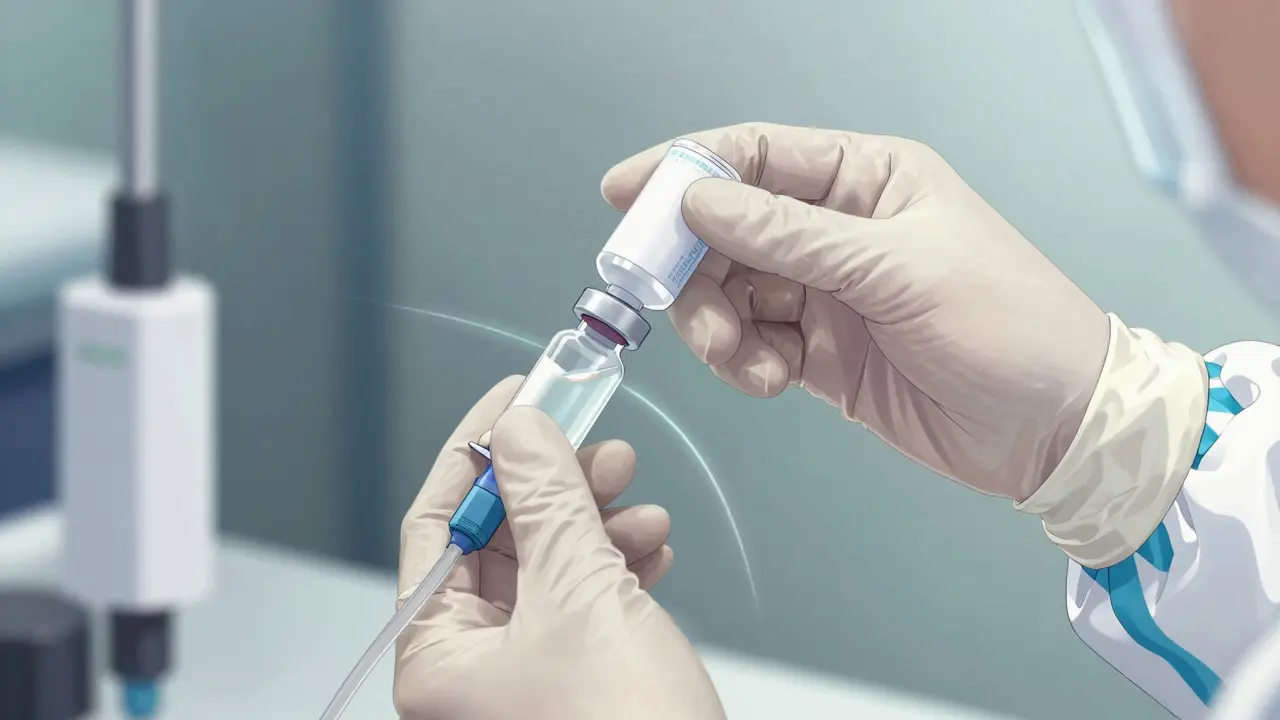
You cannot rely on gloves alone when handling hazardous substances. Studies dating back to the early 1990s showed that contamination can transfer from outer gloves to hands and surfaces if the gear isn't chosen correctly. This is where specific equipment becomes non-negotiable for staff safety.
| Equipment Type | Safety Requirement | Rationale |
|---|---|---|
| Gloves | Chemo-tested double gloves | Prevents permeation from high-risk agents like carmustine |
| Gowns | Impermeable material | Stops liquid drugs from soaking through fabric |
| Eye Protection | Mandatory when splashing is possible | Protects mucous membranes from aerosolized particles |
| Respiratory Masks | Required for spill cleanup or aerosols | Prevents inhalation of volatile compounds |
All personal protective equipment (PPE) is considered contaminated once exposed to the environment where chemotherapy is handled. You can't just wash and reuse them after a minor splash incident. If a glove touches a clean surface after touching a drug bag, that surface is now a hazard zone. This applies equally to the gown. Facilities must invest in Closed-System Transfer Devices (CSTDs) as part of their engineering controls. These devices prevent vapors or liquids from escaping the vial during preparation.
The cost factor is real. Implementing full compliance often requires an initial investment ranging between $18,500 and $25,000 per treatment facility. While that sounds steep, data shows that facilities fully adhering to these protocols see 63% fewer medication errors. That reduction saves money in malpractice claims and prevents severe health outcomes for the workforce.
Managing Risks in the Home Environment
Hospital walls offer engineered safety zones, but cancer treatment increasingly happens in private homes. About 22% of home care incidents involve improper disposal of hazardous waste. Patients and caregivers are the frontline workers here. They need clear instructions on bodily fluid management for 48 to 72 hours after treatment ends.
It is critical to store medication and supplies in child-proof locations immediately. Caregivers report feeling unprepared, especially regarding sharps disposal. Using a formal toolkit designed for home safety reduces these concerns significantly. Even though the home environment lacks the HEPA filtration found in clinics, following strict hygiene protocols minimizes exposure risks for partners and children living in the same household.
Addressing New Threats: Cytokine Release Syndrome
Newer therapies bring new complications. The 2024 standards explicitly address monitoring for Cytokine Release Syndrome (CRS). This condition involves a systemic inflammatory response to certain immunotherapies. Between 2018 and 2022, cases of CRS surged by 300%. Without immediate intervention, mortality rates sit between 12% and 15%.
Facilities must now keep antidotes and specific CRS-directed therapies readily accessible during administration. This means nursing stations cannot just store general emergency kits. They need specialized medication that targets the cytokine storm specifically. Staff training requires demonstrating competency in recognizing early signs like fever spikes or blood pressure drops that precede the crisis.

Staff Competency and Training Requirements
Anyone handling antineoplastic agents must prove they know what they are doing. The standards mandate specific certification training. Initial certification usually takes 8 to 12 hours. After that, annual refreshers of four hours are required to keep skills sharp. Personnel must pass a written exam scoring at least 85% and show observed practical demonstration of safety procedures.
Training gaps are a major source of error. In community cancer centers, rural programs sometimes struggle to afford the full range of engineering controls like CSTDs. Experts argue this creates a two-tiered system where urban patients get safer handling than rural ones. To fix this, organizations are pushing for standardized national certification expected by 2026. Until then, facilities must audit their own staff annually to ensure no lapses in protocol.
Implementation Challenges and Costs
Adopting these standards is a process, not a switch flip. For a medium-sized practice, getting certified typically takes 9 to 12 months. Electronic Health Records (EHR) often cause the biggest headache. About 67% of facilities report needing custom builds to support the multiple verification steps required by law. Custom software development can cost between $15,000 and $40,000 per facility.
Regulatory enforcement is tightening too. Regulatory bodies issued over 140 citations for hazardous drug handling violations in a single recent year, marking a 37% increase from the previous period. Average penalties hit around $14,250 per violation. Ignoring the rules isn't a financial option anymore. Facilities that treat safety as optional risk not just fines, but losing accreditation necessary to receive insurance payments.
Despite the workload, most nurses feel better equipped. Surveys indicate that over three-quarters of oncology nurses have improved confidence after full implementation. The standardized time-out verification process alone reduced near-miss errors by over half. However, some staff complain that the extra verification steps add 10 minutes per patient. Balancing patient throughput with absolute safety remains the constant tension in oncology care.
What is the purpose of the fourth verification step?
The fourth verification step requires two licensed clinicians to confirm patient identity and medication details at the bedside. This specific addition to the 2024 standards aims to stop patient identification errors, which historically caused 18% of chemotherapy-related adverse events.
Do caregivers at home need special safety equipment?
Yes, home caregivers should use chemotherapy-specific spill kits, store meds in child-proof containers, and follow fluid disposal protocols for up to 72 hours post-treatment. Proper sharps disposal containers are essential to prevent needlestick injuries.
Which drugs require double gloving?
High-risk drugs like carmustine and thiotepa require chemo-tested double gloves to prevent chemical permeation through standard latex barriers. Standard guidelines recommend double gloving for all antineoplastic preparations as a baseline safety measure.
How often must staff retrain on safety protocols?
Staff require an initial 8-12 hour training certification. Following this, annual refresher courses lasting four hours are mandatory to maintain competency and adhere to ASCO and ONS safety standards.
What defines a closed-system transfer device (CSTD)?
A CSTD is a mechanical device that prohibits the escape of hazardous drug aerosols or vapors. It connects the vial to the syringe, ensuring no contaminants enter the air during medication preparation, a requirement emphasized in European and US standards.


It is truly tragic how we quantify human safety in dollars. The data regarding malpractice claims sounds appealing on paper yet ignores the soul of the institution. We see these frameworks evolving but do they address the fundamental fear. Nurses spend their careers fearing permeation through skin barriers while administrators count pennies. The twenty thousand dollar investment is negligible compared to a life lost but it feels heavier than that. Standardized national certification by two thousand twenty six seems like a distant dream for rural centers. Geography currently determines the safety of a patient receiving treatment. Urban facilities enjoy CSTD protections while country clinics struggle with basic airflow. This disparity creates a hierarchy of survival based on zip codes alone. We claim progress yet the gap between policy and practice remains wide. Training gaps are cited frequently but never fully resolved in community centers. A written exam score of eighty five percent does not guarantee true competency. Practical demonstration is required yet observed skills often fade during night shifts. Regulatory citations have increased drastically showing a pattern of negligence. Penalties average fourteen thousand dollars per violation which is nothing compared to reputation loss. Accreditations become the real threat when insurance payments stop flowing into accounts. Ignoring the rules is not a financial option anymore but the culture shift is slow. Safety must be absolute rather than optional for those handling hazardous drugs. The constant tension between throughput and absolute safety defines modern oncology care. This balance tips too often toward speed instead of caution. We must demand better than mere compliance with new legal mandates.
That disparity in rural access is a known logistical bottleneck.
It is really important to understand home safety for caregivers. Many families feel unprepared regarding sharps disposal. You cannot rely on standard gloves for bodily fluid management. The forty eight hour window after treatment is critical for partners and children. Proper storage in child proof locations makes a huge diffrence. Even though home environments lack hepa filtration strict hygiene helps minimize risks. Caregivers report feeling overwhelmed without clear instuctions on waste. Using a formal toolkit designed for home safety reduces these concerns significantly. Families need to know that contamination can transfer from outer gloves to hands easily. Education requirements mean patients must document understanding of duration and side effects. We need more support systems for homecare workers who manage this daily. The update standards help protect everyone involved in the process.
Home care protocols are definitely underfunded in many regions but the guidance helps
The gear requirements are super important for keeping staff safe 💪 Wearing impermeable gowns stops liquids from soaking through 🛡️ Double gloves prevent permeation from high risk agents like carmustine 🧤 Eye protection is mandatory when splashing could happen 👁️ Respiratory masks are needed for spill cleanup or aerosols 😷 Contaminated gear means you cant just wash and reuse them after a splash incident 🚫 All ppe becomes contaminated once exposed to the handling environment 📌 Closed system transfer devices prevent vapors escaping the vial 🔒 These protocols save lives and reduce errors significantly ❤️ Staying safe allows us to focus on patient care better 🏥
Absolutely right about the gear specs :) The permeation prevention is key for long term health ((o_O)) We need to normalize wearing full protection everywhere :D It shows respect for the work being done there (^^) Spill kits at home are also a great tip for families :P Safety culture starts with the basics like proper glove choice :) Thank you for highlighting those details (:)
From a compliance standpoint the CSTD implementation is critical for volatile compound mitigation. Our EHR integration requires custom builds to support multi-step verification logic. Current ASCO and ONS joint committee guidelines mandate independent confirmation protocols. Patient verification now includes a fourth step performed at the bedside before administration. This catches identification errors accounting for nearly eighteen percent of adverse events. Automated systems miss nuances that dual clinician sign-off captures effectively. Staff competency requires initial certification lasting eight to twelve hours minimum. Annual refreshers of four hours maintain skill sharpness over time. Facilities must pass written exams scoring at least eighty-five percent. Observed practical demonstration validates physical handling proficiency. Engineering controls like closed-system transfer devices are non-negotiable investments. Initial capital expenditure ranges between eighteen and twenty-five thousand dollars per site. Full adherence correlates with sixty-three percent fewer medication errors reported. Malpractice claims reduction offsets the upfront operational costs quickly. We need robust audit trails documenting every single safety step taken.
This is absolutely vital information!!!!! The accuracy of verification steps cannot be understated!!!! Dual clinician confirmation saves lives directly!!!! We must push for these standards across the board!!!! The cost is worth preventing severe health outcomes!!!! Staff confidence improves massively with proper training!!!! Compliance is not optional anymore!!!!
I think the focus on competency training is fantastic! Ensuring every nurse passes the written exam builds trust. Seeing staff demonstrate safety procedures practically gives me so much hope! We are moving away from simply being careful to verifiable protocols. It empowers the workforce to handle hazardous drugs confidently. The goal of reducing near-miss errors by half is incredibly motivating. Every minute spent verifying patient identity prevents disaster. We celebrate the improvements made by three quarters of oncology nurses. Their confidence after full implementation is something to cherish. Let's keep pushing for consistent national certification by 2026. Rural programs deserve equal access to engineering controls like CSTDs. Together we can create safer environments for everyone involved!
Fantastic optimism indeed but ignoring the reality of budget constraints is naive. Ten minutes per patient adds up to massive delays in care delivery. Throughput remains the constant tension nobody wants to address honestly. Balancing absolute safety with speed is a losing battle for most hospitals. Some staff feel the extra steps burden the workflow unnecessarily. Accreditation loss is a real threat yet ignored in cheerful posts like this. Fines hit around fourteen thousand dollars per violation regardless of good intent.